Lab Research Projects
Establishing a systematic approach to translate human genetic findings of Coronary Artery Disease into novel biology and medicine
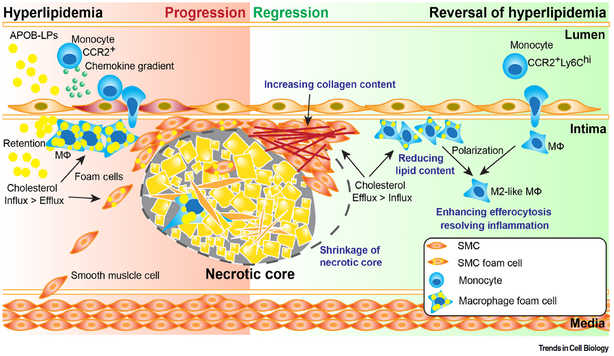
Atherosclerotic coronary artery disease (CAD) represents the leading cause of death worldwide. While human genetic studies have uncovered over 200 loci associated with CAD, a key impediment for translating these genetic findings to clinic practice is the lack of high-throughput functional screening platforms to rapidly ascertain the causal genes linked to cardiovascular phenotypes. Therefore, we seek to combine functional genomics, CRISPR-based high-throughput screens, computational biology and animal models to achieve three fundamental goals: (1) to identify novel causal genes linked to CAD; (2) to discover novel mechanisms regulating atherosclerotic plaque progression and regression; (3) to use the mechanistic insights to develop new therapies for the treatment of CAD.
Atherosclerotic coronary artery disease (CAD) represents the leading cause of death worldwide. While human genetic studies have uncovered over 200 loci associated with CAD, a key impediment for translating these genetic findings to clinic practice is the lack of high-throughput functional screening platforms to rapidly ascertain the causal genes linked to cardiovascular phenotypes. Therefore, we seek to combine functional genomics, CRISPR-based high-throughput screens, computational biology and animal models to achieve three fundamental goals: (1) to identify novel causal genes linked to CAD; (2) to discover novel mechanisms regulating atherosclerotic plaque progression and regression; (3) to use the mechanistic insights to develop new therapies for the treatment of CAD.
Elucidating molecular mechanisms regulating lipotoxicity in Nonalcoholic Steatohepatitis (NASH)
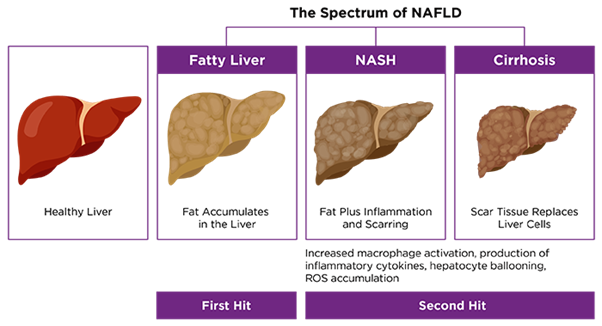
NASH is a more serious form of nonalcoholic fatty liver disease (NAFLD) that is under global prevalence. NASH is a chronic inflammatory liver disease caused by excessive accumulation of fat in hepatocytes. Numerous therapeutic strategies targeting different pathological processes of NASH, such as fat accumulation, inflammation and fibrosis, are currently in clinical testing, yet there are no approved therapies. This highlights the urgent need to gain deeper understanding of pathogenesis of NASH for the future development of new therapies.
Our lab combined human genetics, CRISPR-based high-throughput screens, computational biology and animal models to discover novel genes/pathways regulating liver lipotoxicity and inflammation.
NASH is a more serious form of nonalcoholic fatty liver disease (NAFLD) that is under global prevalence. NASH is a chronic inflammatory liver disease caused by excessive accumulation of fat in hepatocytes. Numerous therapeutic strategies targeting different pathological processes of NASH, such as fat accumulation, inflammation and fibrosis, are currently in clinical testing, yet there are no approved therapies. This highlights the urgent need to gain deeper understanding of pathogenesis of NASH for the future development of new therapies.
Our lab combined human genetics, CRISPR-based high-throughput screens, computational biology and animal models to discover novel genes/pathways regulating liver lipotoxicity and inflammation.
Improving prime editors for the treatment of severe monogenic liver disease
Liver plays a key role in regulating multiple complex metabolic pathways and hepatocytes are affected in many inherited genetic diseases. Generally, liver monogenetic diseases are rare, but many of them are life-threatening and usually require organ transplantation as a curative approach. However, liver transplantation is often limited due to the shortage of donors, as well as its association with mortality/ morbidity in the patients. CRISPR-based genome editing technologies hold tremendous promise for the treatment of monogenic liver disorders as they have the potential to permanently and precisely modify disease-causing genes in the liver of patients.
In the CRISPR toolbox, prime editor represents the most versatile genome editing tool that enables a variety of sequence modifications on genome, including base substitutions, deletions, and insertions, without the requirement for a DSB or exogenous donor DNA templates. Our lab seeks to further engineer the current PE thereby enhancing its editing efficiency and to employ clinically approved lipid nanoparticle (LNP) to deliver primer editor into mouse liver thereby interrogating its editing efficiency in vivo.
Liver plays a key role in regulating multiple complex metabolic pathways and hepatocytes are affected in many inherited genetic diseases. Generally, liver monogenetic diseases are rare, but many of them are life-threatening and usually require organ transplantation as a curative approach. However, liver transplantation is often limited due to the shortage of donors, as well as its association with mortality/ morbidity in the patients. CRISPR-based genome editing technologies hold tremendous promise for the treatment of monogenic liver disorders as they have the potential to permanently and precisely modify disease-causing genes in the liver of patients.
In the CRISPR toolbox, prime editor represents the most versatile genome editing tool that enables a variety of sequence modifications on genome, including base substitutions, deletions, and insertions, without the requirement for a DSB or exogenous donor DNA templates. Our lab seeks to further engineer the current PE thereby enhancing its editing efficiency and to employ clinically approved lipid nanoparticle (LNP) to deliver primer editor into mouse liver thereby interrogating its editing efficiency in vivo.